Fish
Quantitative - Qualitative
Deductive - Inductive
Individual - System - Global
Past - Present - Future
In short: Bankside counts, hook and lining and lift, throw and push netting are methods to census fish populations. They differ in their suitability for different sizes, feeding habits and densities of fish as well as the environmental conditions they are suitable for. Moreover, as a side effect, they cause different magnitudes of damage to fish that have been caught and released (Table 1).
Table 1. Suitability of bankside counts, hook and lining and lift, throw and push netting for census of fish populations.
| Bankside counts | Hook and lining | Lift, throw and push netting | |
| Size of fish | large | large | small |
| Feeding habit | all | predatory | all |
| Density | high | low | all |
| Depth of water | shallow | deep | shallow |
| Water conditions | calm | all | still or slowly flowing |
| Damage to fish | none | big | small |
Contents
Bankside counts
What the method does
Bankside counts are a good way to census conspicuous fish in pools and slow-moving, clear freshwater streams (Figure 1). It enables you to count fish in a stretch of water without catching them. First, you need to divide the stretch of water to be surveyed into adjacent non-overlapping sections by using landmarks on the shore. Every section needs to be completely visible from a certain vantage point at the riverside.

The observer, wearing polarising sunglasses to reduce the glare of the water surface and dull clothing, stands at the vantage point without moving for at least five minutes so the fishes’ behaviour is not impacted by the presence of the observer anymore. After that time, the observer counts all the fish that are visible within this section. During the process of counting, the observer should not move to prevent fish from fleeing. Afterwards, the observer moves away from the bankside before moving on to the next section and stands at the next vantage point.
It is possible to calculate the density of fish individuals after measuring the length and width of the river section.
Strengths & challenges
This method is cheap, easy to conduct and does not put the fish under a lot of stress. Parameters such as sex, size of the fish and many environmental measures such as water depths, substrate type and vegetation can be recorded. The biggest challenge is that fish are very aware of human presence and change their behaviour immediately when they are slightly disturbed. They take a relatively long time to resume their usual behaviour after being disturbed. Therefore, a high inter-observer variability is reported for this method (Hankin and Reeves 1988).
Hook and lining
What the method does
Hook and lining is a method to census large, predatory fish that occur at low density. It is suitable for fast-flowing rivers, deep lakes and in the sea. For catching predatory fish, live invertebrates, such as worms, are added to a hook. The bait can also be imitating worms, flies, small fish etc. The line to be used can either be short and directly attached to the hand or long and strung to a pole or rod. By doing so, the researchers do not have to get as closely to the fish which prevents the fish from seeing them. The line is then thrown out into the water, either from the shore or from a boat. By periodically pulling on the line, the researcher can mimic the movements of a living invertebrate and thereby, increase the chances of catching a fish. As soon as the line straightens, it should be pulled up to embed the hook into the fish’s mouth. As soon as the fish reaches the water surface, it should be caught with a net and the hook should be removed carefully. Until the sampling is done, fish can be kept in keepnets or water filled containers. When the fishes are marked and later recaptured, it is possible to generate quantitative information on population parameters. Otherwise, catch per unit effort statistics are generated for particular targeted groups or species. Before trying to census fish with this methodology, it is recommended to get advice from local fishers on which baits to use and where to fish. Additionally, it is helpful to design tournaments of local anglers as a means to get enough labor force for a fish census.
Strengths & challenges
The strength of this method is that it is cheap and can be used also for low densities of fish. Moreover, it is useful in habitats that cannot be sampled with other methods. However, this method is highly biased as it is connected to a context-specific selectivity and therefore cannot be used for monitoring entire fish communities. The bait used will determine the species that are caught. Moreover, the success of this method is strongly dependent on the environmental conditions such as weather, water temperature and timing.
Lift, throw and push netting
What the method does
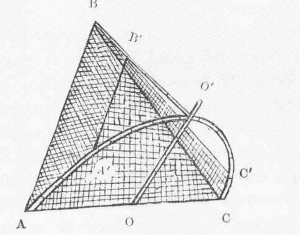
Lift nets are scoop nets that are inserted below the water surface and brought up sharply (Figure2). A special case of lift nets are buoyant nets which are naturally floating but attached to a heavy frame that keeps the net under water. The glue that attaches the net to the frame dissolves over time, so that the net moves to the surface and catches fish on its way. As a glue, dissolving tapes can be used.
Cast nets are circular nets with weights around the perimeter (Figure 3). The net is thrown from a bank or from a boat and then hauled by pulling the central line. When the net is hauled, it collapses, and the fish are tangled in the meshes. This method takes a lot of experience to be conducted correctly. Otherwise, it is hard to throw the net in a way that maximises the area sampled.
Push nets consist of a pocket-shaped net that is attached to a D-shaped frame (Figure 4). The frame is pushed over the ground in shallow water. To make pushing easier, some push nets even have rollers. Netting with push nets can be standardized by always taking the same number of paces while pushing.


Strengths & challenges
Although these methods take some practice, lift, throw and push netting are easily repeatable and cheap. However, these methods can become very labor intensive if a lot of samples should be taken. For highly vegetated habitats, lift and cast netting are recommended, whereas push netting can only be conducted in sparsely vegetated waters with hard ground.
Normativity
- A major bias for bankside counts is that big and colourful fish can be seen more easily than small or dull fish. Therefore, the percentage of male fish is mostly overrated. As fish in a certain age group might tend to seek more cover than fish in different age groups, age bias is also possible. - The biggest problem with hook and lining is that fish are damaged by biting the hook. Although the hook can be removed without bigger issues in most cases, not all fishes that were caught survive. Fish most likely die after catch and release due to intracellular acidosis within the muscles after a long fight and due to wounds from big hooks (Gargan et al 2006; Wood et al. 1983). Therefore, small and barbless hooks are suggested to lessen injury to the fish (Sutherland 1996). - For lift and cast netting, little is known about their selectivity and efficiency. Therefore, the results could potentially be strongly biased. All three methods only capture a very small area, so that only fish are caught which are small and rather slow. Catching fish with a net puts them under significant stress but does not harm them physically if they are treated carefully and only touched with wet hands (Sutherland 1996). After measuring and counting, these fish can be released into the water. Air exposure should be kept as short as possible.
Outlook
The biggest issues of the above mentioned methods are that their results are strongly biased towards fish with certain functional traits and for lift and cast netting, it is even unclear how specific the methods are. Moreover, except for bankside counts, they cause damage and/ or stress to the fish. In the future, the trend towards genetic analyses will therefore most likely continue as the new methods are developed further. For instance, environmental DNA meta barcoding (“eDNA”) is a non-invasive method that “harvests” DNA fragments from the environment (Aglieri et al. 2020). Therefore, it is not biased towards fish with certain functional traits and also detects rare and low-abundance species that would be left out using the above mentioned traditional methods. Currently, this method is still rather expensive and a laboratory with devices for DNA barcoding is needed. However, in the future, this method is likely to become more affordable as it is used more extensively. Fish telemetry is another method that has been increasingly used in the last 15-25 years (Thorstad et al. 2015). Telemetry is “the system of determining information about an animal through the use of radio signals from or to a device carried by the animal” (Gutema 2015). The most used methods for fish telemetry are radio and acoustic transmitters, data storage tags (DST), pop-up satellite archival tags (PSAT) and passive integrated transponder tags (PIT-tag) Telemetry is applied to record information on movements, position and measurements of environmental parameters (Thorstad et al. 2015). This information is needed to better protect and manage fish populations by assessing the effects of hydropower, water pollution and catch-and-release-angling, among others (Thorstad et al. 2015). Therefore, tags are attached to the fish by surgical implantation into the body, external attachment, and gastric insertion through the mouth. Negative effects on the individual such as inflammation, infections, decreased swimming performance and increased mortality can negatively impact the study results. Therefore, and for ethical reasons, handling of fish should cause as little damage as possible. As tags are getting smaller and causing less harm through technical development, the trend towards telemetry as a method for fish census is likely to continue (Thorstad et al. 2015).
Key publications
Theoretical:
Sutherland, W. J., Editor (1996). Ecological Census Techniques - a Handbook. P. 178-180. Cambridge University Press.
Empirical:
Hankin, D. G., & Reeves, G. H. (1988). Estimating total fish abundance and total habitat area in small streams based on visual estimation methods. Canadian journal of fisheries and aquatic sciences, 45(5), 834-844.
References
(1) Aglieri, G., Baillie, C., Mariani, S., Cattano, C., Calò, A., Turco, G., Spatafora, D., di Franco, A., di Lorenzo, M., Guidetti, P., & Milazzo, M. (2021). Environmental DNA effectively captures functional diversity of coastal fish communities. Molecular Ecology, 30(13), 3127–3139. https://doi.org/10.1111/mec.15661
(2) Gargan, P. G., Stafford, T., Økland, F., & Thorstad, E. B. (2015). Survival of wild Atlantic salmon (Salmo salar) after catch and release angling in three Irish rivers. Fisheries Research, 161, 252–260. https://doi.org/10.1016/j.fishres.2014.08.005
(3) Hankin, D. G., & Reeves, G. H. (1988). Estimating total fish abundance and total habitat area in small streams based on visual estimation methods. Canadian journal of fisheries and aquatic sciences, 45(5), 834-844.
(4) Sutherland, W. J., Editor (1996). Ecological Census Techniques - a Handbook. P. 178-180. Cambridge University Press.
(5) Thorstad, E. B., Rikardsen, A. H., Alp, A., & Økland, F. (2013). The use of electronic tags in fish research–an overview of fish telemetry methods. Turkish Journal of Fisheries and Aquatic Sciences, 13(5), 881-896.
The author of this entry is Anna-Lena Rau.